
Research
Molecular characterisation of microRNA-directed responses to abiotic stress in plants
During the course of my PhD thesis research on plant molecular genetics at CSIRO Plant Industry Canberra, I became fascinated in the then emerging field of RNA silencing: an endogenous mechanism of gene expression regulation that occurs at the RNA level. In plants, RNA silencing is directed by various species of small RNA to regulate gene expression during all phases of vegetative and reproductive development. More recently, a specific species of small RNA, the microRNA (miRNA) species of small RNA, has been demonstrated across a wide range of evolutionary unrelated plant species, to direct a central role in altering the gene expression changes required for a plant to mount a defence response against an invading pathogen or to adapt to a changing environment.
At the University of Newcastle, we aim to identify the miRNAs responsible for driving the gene expression changes required by a plant to adapt to environmental stress, with a particular interest in the identification of miRNAs responsive to the abiotic stresses central to Australian cropping agriculture, drought stress, heat stress and salt stress. We initially base our studies in the genetic model plant species Arabidopsis thaliana (Arabidopsis). That is, we expose Arabidopsis to environmental stress to identify the stress responsive miRNAs of interest, and then via a molecular approach, we generate new Arabidopsis plant lines to determine if the introduced molecular modifications provide these plant lines with an added degree of tolerance to the applied stress. miRNAs, and the target genes under miRNA-directed expression regulation, have been highly conserved across plant evolution. Taking advantage of this, a future goal of the Eamens Lab is to translate the research findings made in Arabidopsis to agronomically important crop species.
In collaboration with the Grof Lab, we have also recently developed a transformation platform in the C4 monocot grass, Setaria viridis. Development of this platform, and of a suite of other S. viridis specific molecular tools, will allow for the molecular modification of this newly identified C4 model. C4 plants have a highly specialised leaf anatomy compared to that of C3 leaves, therefore; we aim to use S. viridis to molecularly characterise miRNA-directed adaptive responses to environmental stress in C4 plants, in parallel to those studies we currently undertake in Arabidopsis, a C3 plant.
If taking a molecular approach to functionally characterise miRNA-directed adaptation to environmental stress in plants sounds like an exciting research career direction to you, then please do not hesitate to contact me for further information.
Andrew Eamens: andy.eamens@newcastle.edu.au
Discovering Mechanisms Regulating Transfer Cell Development
A significant constraint on plant and crop productivity are rates at which nutrients (carbon and nitrogenous compounds and minerals) are transported across plasma membranes (PMs) of cells that acquire nutrients from the soil solution and of cells that load/unload vascular pipelines that convey acquired nutrients from sources (net nutrient exporters; e.g., fully-expanded leaves) to sinks (net nutrient importers; e.g., developing seeds and fruits). Nutrient transport rates per cell are the product of their PM transporter densities (activity) and PM surface areas in which the nutrient transporters are embedded.
Transfer cells (TCs) are the most evolutionarily advanced cell type specialized to support high rates of nutrient transport across their PMs. The transport capacity of TCs is conferred by an amplified PM surface area, enriched in nutrient transporters, arrayed on an invaginated wall labyrinth, commonly polarized to the direction of nutrient transport. The degree of wall labyrinth invagination, and hence PM amplification, is determined by the number of repeating fenestrated layers of vertically-oriented wall ingrowth papillae (WIs; Fig. 1).
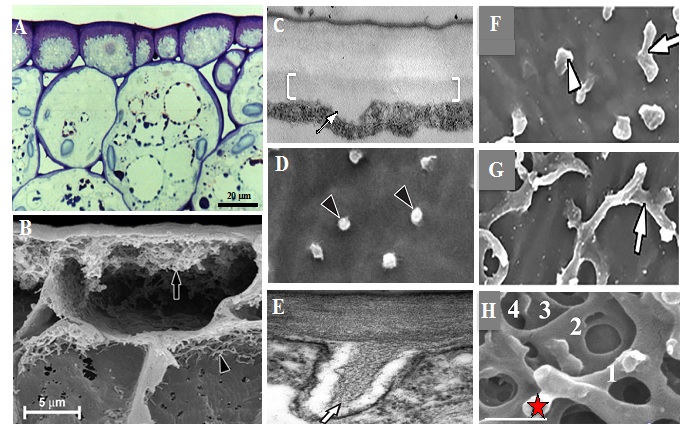
Fig. 1. A. Light micrograph of a transverse sectionof epidermal transfer cells (etc)of a Vicia faba cotyledonillustrating the thick band of wall labyrinth (purple) polarized to the outer wall of each cell. B. Scanning electron micrograph of a freeze fractured cotyledon with the cytoplasm removed showing the reticulate invaginated wall labyrinth (black arrow). C - H. Wall labyrinth assembly in epidermal cells of cultured V. faba cotyledons. C, D. Transverse (C) and cytoplasmic face (D) views of the uniform wall (brackets in C) from which WI papillae arise (darts in C, D). E. Transverse section of a WI papilla showing electron dense cellulose microfibrils (arrow) ensheathed by an electron translucent band of callose. F, G. WI papillae ultimately bend (F, dart) and fuse (G) to form the first fenestrated wall layer (G - arrow) from which another set of WI papillae arise (H-star) H. Cytoplasmic view of a multi-layered fenestrated network of a mature wall labyrinth (fenestrated layers are numbered; star indicates emerging WI papilla). Bar = 1 µm in C, D, F, G; 2.5 µm in E, H.
Thus, discovering mechanisms regulating TC development opens up a relatively unexplored pathway to understand, and hence manipulate, nutrient transport rates in plants and hence potentially improve crop yields. Our research program is being progressed in two phases:
- Currently our focus is to discover signalling cascades, and the down-stream machinery, responsible for assembling the TC wall labyrinth.
- A parallel phase of enquiry, yet to be activated, is to elucidate mechanisms regulating the expression of key transporter proteins and their subsequent targetting to, and embedding in, the amplified PM.
An unique experimental model to study transfer cell development
Progress in studying TC biology has been impeded by impaired experimental access to these specialized transport cells that characteristically occur in low numbers embedded deep within tissue matrices. This impasse has been breached by our development of a TC induction system using cultured V. faba cotyledons. The system offers access to a large population of uncommitted epidermal cells that, upon transfer to culture, are synchronously induced to rapidly (h) trans-differentiate to a proven TC morphology and enhance nutrient transport capacity (Fig. 2). Exposure of cotyledons to pharmacological blockades of the inductive signals creates opportunities to experimentally perturb trans-differentiation at specific developmental stages of wall labyrinth assembly. Peels of trans-differentiating epidermal TCs (ETCs) can be obtained in sufficient quantities for cell-specific gene expression studies and to quantitatively determine the extent of wall labyrinth development by SEM/FESEM using our dry-cleave method. The former has permitted the generation of valuable comprehensive transciptomes of ETCs at specific stages of wall labyrinth assembly as well as transcriptomes of ETCs in which key signals have been pharmacologically blocked. Live-cell imaging of fluorescent-tagged proteins and their signals is available through spinning disc and confocal microscopy. This capacity will be strengthened by developing a transient transformation system that currently has reached proof of concept with colleagues at La Trobe University. Through an international collaboration, we have access to a V. narbonensis stable transformation system to characterize the functional significance of putative key protein players as components of the regulatory mechanism controlling ETC development.
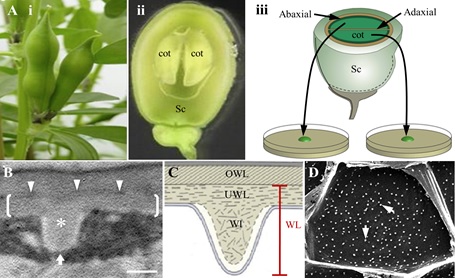
Fig. 2. A.Schematic of V. faba cotyledon culture system illustrating (i) pods prior to harvest, (ii) a seed, cut in half longitudinally, showing the coat (Sc) enclosing two “sister” cotyledons (cot), (iii) “sister” cotyledons removed from the seed coat, are separated and placed adaxial epidermal surface down on the culture medium. B, C. Transmission electron micrograph (B) and diagrammatic representation (C). of a transverse section of the outer periclinal wall of a trans-differentiating adaxial epidermal transfer cell. Brackets in B mark the uniform wall layer (UWL) deposited on the original wall (OWL) and demarcated by an electron-dense band (darts in B); a wall ingrowth (WI) papilla is marked with an asterisk in B. D. Scanning electron micrograph of WI papillae deposition at the cytoplasmic face of the outer periclinal wall of adaxial epidermal cells. Each white dot represents a single WI papilla (arrows). The white arrow in B indicates the direction of imaging of the cytoplasmic face. Bar, 300 nm in B and 10 μm in D.
Current understanding of mechanisms regulating wall labyrinth assembly using cultured V. faba cotyledons
Given that TCs are located within the plant body at specific sites of intense nutrient transport, combined with the complexity of their wall labyrinth assembly (Fig. 1), we envisage a multiple-layered regulatory system controlling TC development. Broadly we propose the system comprises an interplay between a cohort of primary signals that switch on a gene network that drives formation of a central signalling hub which in turn regulates temporal expression of cell wall biosynthetic machinery involved in, and provides positional information for, the assembly of the intricate wall labyrinth.
Our cell and molecular studies of epidermal TC development in cultured V. faba cotyledons have discovered the following elements of the proposed multi-layered regulatory system. These have been organized into a working model (Fig. 3). A complex cascade of primary signals funnel to transcriptionally activate ethylene biosynthesis and a signalling pathway that elicits an extracellular reactive oxygen species (ROS) signal polarized to the outer periclinal domain of the ETCs. The latter initiates cell wall biosynthesis and directs polarized assembly of the uniform wall layer independent of the cytoskeleton. Cross-talk between the ethylene/ROS signals orchestrates formation of inward-directed plumes of elevated cytosolic Ca2+ ([Ca2+]cyt) that define punctate domains at which WI papillae arise from the uniform wall layer. Genes encoding proteins contributing to generating the ROS and Ca2+ signals along with those synthesizing the callose sheath and cellulose microfibrils have been identified. We envisage these key proteins undergo rapid and profound temporal and spatial changes in the outer periclinal domain of the trans-differentiating epidermal cells to ensure the ordered sequential assembly of the structurally-complex wall labyrinth.
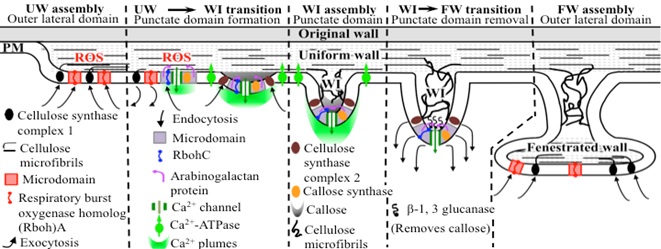
Fig. 3. A working model of mechanisms proposed to regulate the spatiotemporal dynamics of key plasma membrane (PM) proteins responsible for assembling the wall labyrinth of epidermal transfer cells of cultured V. faba cotyledons.
The working model (Fig. 3) provides the conceptual framework for formulating current and future projects designed to discover the mechanisms regulating wall labyrinth assembly. Many questions remain, for example: What is the role of sterol-enriched plasma membrane domains? How is the ROS signal generated?
Enquiries:
Professor Tina Offler
Professor John Patrick
Surge into new agribusiness medicinal Cannabis venture
Recent legislative changes in Australia have provided a remarkable opportunity to participate in the development of a new, international scale industry estimated to be worth 55 billion dollars globally by 2025. The Centre for Plant Science through Prof Grof, has engaged with an innovative medicinal Cannabis company, CannaPacific, based in northern NSW. An initial engagement with CannaPacific was catalysed by the Faculty of Science via the Matched Industry Funding Scheme. Through CannaPacific, the Centre has direct links to a major pharmaceutical company. Extensive R&D opportunities in medicinal Cannabis are on the horizon for the Centre, through the conduit provided by an agreement signed between the University of Newcastle and CannaPacific.
Generation of new varieties of Cannabis plants
The initial project is focused on developing and delivering to the Australian medicinal Cannabis industry a series of unique, bespoke medicinal Cannabis varieties. The introduction of novel traits into commercial cultivars of Cannabis, or other agricultural crops, is reliant upon forward or reverse genetic strategies. A reverse genetics approach following introduction of random mutagenic lesions in DNA, will create unique changes in the secondary metabolite profile of Cannabis, likely impossible through conventional breeding practices. To date, directed anthropogenic selection and breeding of Cannabis has primarily favoured traits associated with industrial hemp fibre, hempseed and illicit drug uses
High throughput screening of mutant Cannabis plants
In order to identify the plant lines that possess the capacity to accumulate unique phytocannabinoids, many thousands of plants will have to be screened. The key to such an approach is the development and application of novel, high throughput screening methods. As part of this project, a unique hand-held screening tool is under development by CannaPacific in collaboration with CSIRO.
Detailed characterisation of unique varieties of Cannabis using a range of –omics tools, including genomics, methylomics, transcriptomics, metabolomics and proteomics
Once identified, unique varieties will be subjected to detailed characterisation. The impact of the mutagenic change will be investigated using leading edge tools developed by an Industry partner of CannaPacific, Xing Technologies. The genetic changes underpinning the phenotypic and metabolic changes identified during the screening phase will be determined, providing a powerful platform for unravelling the metabolic complexity underpinning cannabinoid synthesis, leading both to further fundamental discoveries.
CannaPacific Australia
Plant Growth Facility

Enquiries: Professor Christopher Grof
Cell and molecular biology of specialised plant cell walls
Our research focuses on understanding the mechanisms associated with building specialised cell wall structures in phloem parenchyma transfer cells in Arabidopsis, and phi thickening-containing cells of Brassica roots.
Transfer cells in Arabidopsis (McCurdy and Collings)
Transfer cells (TCs) build highly polarised cell wall ingrowths which serve to amplify plasma membrane surface area and hence maximise the nutrient transport capacity of these cells. Apart from their important role in enhancing nutrient transport in plants, and thus influencing crop yield, the deposition of highly polarized wall ingrowths represents a fascinating example of localised wall deposition in plant cells. Our current research on TCs uses the model species Arabidopsis thaliana (Arabidopsis): the enormous genetic and bioinformatics resources available in this species allow us to investigate the molecular regulation of cellular processes leading to TC development. In Arabidopsis, phloem parenchyma (PP) cells in vascular tissue of leaves and leaf-like organs develop wall ingrowths to become PP TCs, performing an important role in phloem loading of photoassimilates into cells of the sieve element/companion cell complex for export to developing tissues and organs. Using a novel fluorescence staining procedure in combination with confocal and super-resolution microscopy, we have mapped PP TC development across cotyledon and leaf development and used this information in combination with RNA-sequencing to identify members of the NAC and MYB gene families that likely participate in the control of PP TC development in Arabidopsis. Since PP TCs play important roles in loading of sugars into the vascular pipeline of source leaves for delivery to developing sink tissues, we are also investigating signals, especially sugars, which may be involved in directing the development of PP TCs.
A major advance in understanding phloem parenchyma transfer cell development in Arabidopsis is our recent discovery that this process represents a novel trait of vegetative phase change, or heteroblasty, in Arabidopsis. The genes and microRNAs that regulate vegetative phase change sit at the interface of vegetative and floral transition, thus providing new opportunities to discover regulatory networks controlling the development of PP TCs.
Phi thickenings in roots of the Brassicaceae (Collings and McCurdy)
Phi thickenings are thickened secondary cell wall bands that are formed around root cortical cells in locations where primary cell walls would normally only be expected. These phi thickenings have been identified in a range of species from gymnosperms to angiosperms, but their function within the root remains unknown. As phi thickenings are not known to occur in the model plant Arabidopsis thaliana, we have instead focussed our research into phi thickening development in the primary root of two agriculturally-important and closely related species, Brassica napus (canola, rapeseed) and Brassica oleracea (broccoli, cauliflower, cabbage and related cultivars). Our research has demonstrated that these phi thickening bands can be rapidly induced by water stress, and that there are wide variations in thickening induction in response to this water stress between different cultivars. Current research is focussed on confirming the functions of phi thickenings within the Brassica root, and identifying the cellular and molecular pathways through which these enigmatic structures develop.
Enquiries


Professor Yong-Ling Ruan focuses his research on identifying molecular and biochemical bottlenecks that limit nutrient allocation to, and utilization within, major organs for improving plant fitness, fertility and food and fibre production. He is a member of ARC College of Experts and Director of Australian-China Research Centre for Crop Improvement. He serves on editorial boards of five international scholarly journals including Molecular Plant, Journal of Plant Physiology and Frontiers in Plant Science.
The Ruan lab aims to:
(i) Elucidate mechanisms by which sugar metabolism & transport regulate plant development;
(ii) Identify regulatory genes that control carbon nutrient distribution from subcellular to whole plant levels;
(iii) Dissect molecular networks underpinning resource partitioning during evolution & domestication.
His ultimate goal is to exploit the knowledge gained from the above programs to develop novel solutions to improve crop productivity and stress tolerance through translational research.
With funding support from ARC and other national and international bodies, Prof Ruan has led his team to a sustained success with research findings and insights published in top journals in Plant Science such as Plant CellandMolecular Plant and in multidisciplines including Nature Genetics and PNAS. For more details please visit: https://www.newcastle.edu.au/profile/yong-ling-ruan
The Ruan group has:
(1) Identified genetic bottlenecks limiting nutrient input into meristematic organs and established a “Ready-Set- Grow” model that provides novel insights into the control of seed & fruit set;
(2) Discovered critical roles of invertase-mediated sugar metabolism & signalling in establishing male & female fertility & cell patterning or growth;
(3) Uncovered (a) the coordinated roles of plasmodesmata (PD) & sugars and K+ transporters in cell expansion and (b) a novel mechanism controlling PD function via callose turnover and sterol homeostasis;
(4) Elucidated evolution models of invertases and related proteins, in which CWINs were co-evolved with seed plants for phloem unloading and reproduction, whereas cytosolic invertases were ancient and steady for maintaining cytosolic sugar homeostasis and signalling.
(5) Demonstrated that elevation of endogenous invertase activity improves leaf longevity & seed yield & confers heat tolerance for fruit set; while enhancing sucrose synthase expression enhanced seedling vigor and cotton fibre yield.
The quality and impact of Prof Ruan's research has been internationally recognized as evidenced by, for example, having written multiple invited articles for world renowned Annual Review of Plant Biology and Trends in Plant Science, ranked first and second, respectively, among all 220+ Plant Biology journals. He has organized and/or chaired many international conferences/ symposia and given invited talks in top institutes worldwide including, for example, University of Cambridge, Cornell University, Max Planck Institute of Molecular Plant Physiology, Friedrich-Alexander University, Ludwig- Maximilian University of Munich, Swedish University of Agricultural Sciences, Swiss Federal Institute of Technology, Peking University and Tokyo University.
Prof Ruan has been awarded the prestigious Peter Goldacre Medal from Australian Society of Plant Scientists, Distinguished Young Scientist Award from National Science Foundation of China and Kuan-PiuVisiting Chair Professorship from Zhejiang University.
Please contact yong-ling.ruan@newcastle.edu.au if you are interested in exploring research collaborations or joining his team as honours or PhD students or postdoctoral fellows.
The University of Newcastle acknowledges the traditional custodians of the lands within our footprint areas: Awabakal, Darkinjung, Biripai, Worimi, Wonnarua, and Eora Nations. We also pay respect to the wisdom of our Elders past and present.
